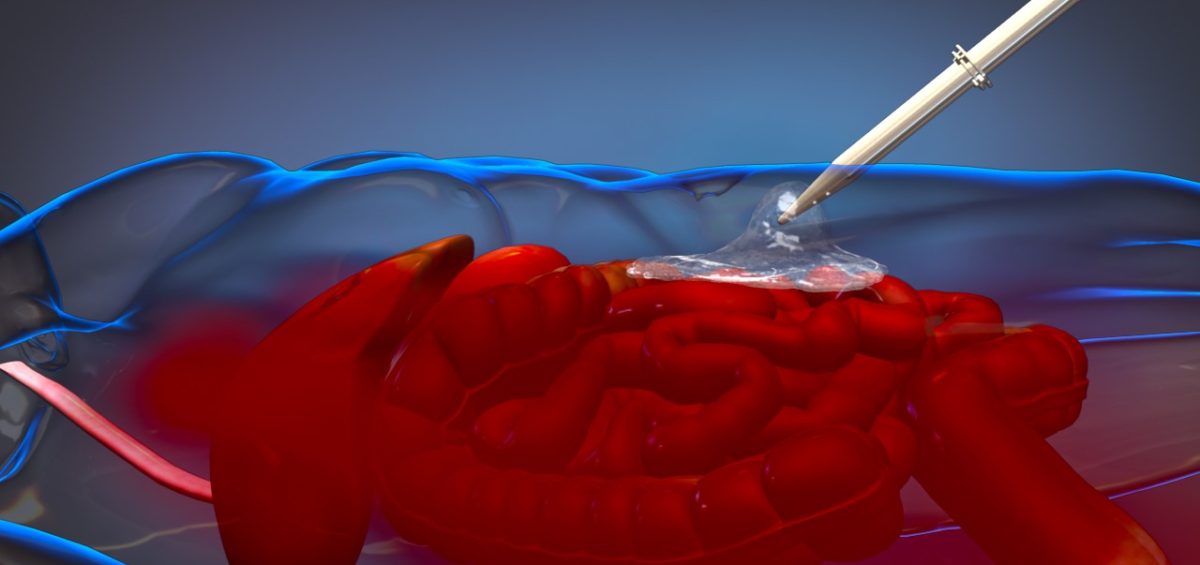
For 10 years, David King, MD, trauma and acute care surgeon in the MGH Trauma Center, and an engineering team have been working to create ResQFoam, a self-expanding polyurethane trauma foam designed to slow or stop internal bleeding in severely injured patients.
ResQFoam is injected into the body, and it quickly expands to fill the abdominal cavity, applying pressure that may stabilize the patient. King says when the patient reaches an operating room, the foam is easily removed.

Dr. King recently received FDA approval to launch the REVIVE Trial, a clinical trial to test the foam as a way to help patients with severe internal bleeding survive long enough to undergo surgery.
“In the history of medicine, the only way to stop internal bleeding has been through surgery,” King says. “With noncompressible abdominal hemorrhage following trauma, you can’t put pressure on the bleeding like you can with other body parts. This foam temporarily stops the bleeding, allowing critical time for the patient to make it to the operating room for surgery.”
Due to the severe nature of the injuries that the foam is designed to treat, the study team has received Exception From Informed Consent (EFIC) status from the FDA. This means the foam can be used to treat qualifying trauma patients without obtaining consent as long as they have not previously opted out of the study.
“This study is for people who are so gravely injured they are likely to not survive, or will have a dismal outcome,” says King. “We’re not talking about the child who falls at soccer practice and comes in with a minor spleen injury. These are the most horrific injuries where the human body does not do well.”
For the next few months, King and his colleagues are reaching out to the community to share information about the clinical trial. “Part of this process is establishing a dialogue with the community and vetting what we think we have already figured out,” King says.
The research team invites you to share your opinions about this upcoming research study that will involve Exception from Informed Consent rules. Please complete this brief questionnaire which will help them better understand how the community feels about this EFIC process.
You can also join the discussion by registering for an upcoming focus group. Contact MGHREVIVETRIAL@gmail.com or 617-643-2440 to learn more.
Individuals who want to opt out of the clinical trial can request a special bracelet. More information on the opt-out process can be found at resqfoam.com. “It is incumbent on us as ethical researchers to make opting out easy,” says King.
King says he hopes ResQFoam will save lives at Mass General, in hospitals throughout the country, on the battlefield and at any point of a trauma.
About the Mass General Research Institute
Research at Massachusetts General Hospital is interwoven through more than 30 different departments, centers and institutes. Our research includes fundamental, lab-based science; clinical trials to test new drugs, devices and diagnostic tools; and community and population-based research to improve health outcomes across populations and eliminate disparities in care.
Support our Research






Leave a Comment