
Jouha Min, PhD, is a postdoctoral fellow in the Center for Systems Biology at Massachusetts General Hospital. Her poster, Point-of-Care Sepsis Diagnostics, was among the winners at this year’s Mass General Scientific Advisory Committee poster session.
Min, along with her Mass General colleagues Ben Coble, Jongmin Park, Hyungsoon Im, Cesar M. Castro, Filip K. Swirski, Hakho Lee, and Ralph Weissleder from the Center for Systems Biology, and Hui Zheng from the Biostatistics Center, as well as Maria Nothing and Georg F. Weber from the University Hospital of Erlangen, have developed a device that can rapidly diagnose sepsis.
We caught up with Min to ask her more about this research.
1) What problem(s) are you addressing with this research?
Sepsis is an often-fatal condition that arises when the body launches an overwhelming immune response to an infection that causes more damage to the body than the infection itself.
A critical unmet need in combating sepsis is the lack of accurate early biomarkers that can alert clinicians to this potential life-threatening situation and allow them to take preventative action.
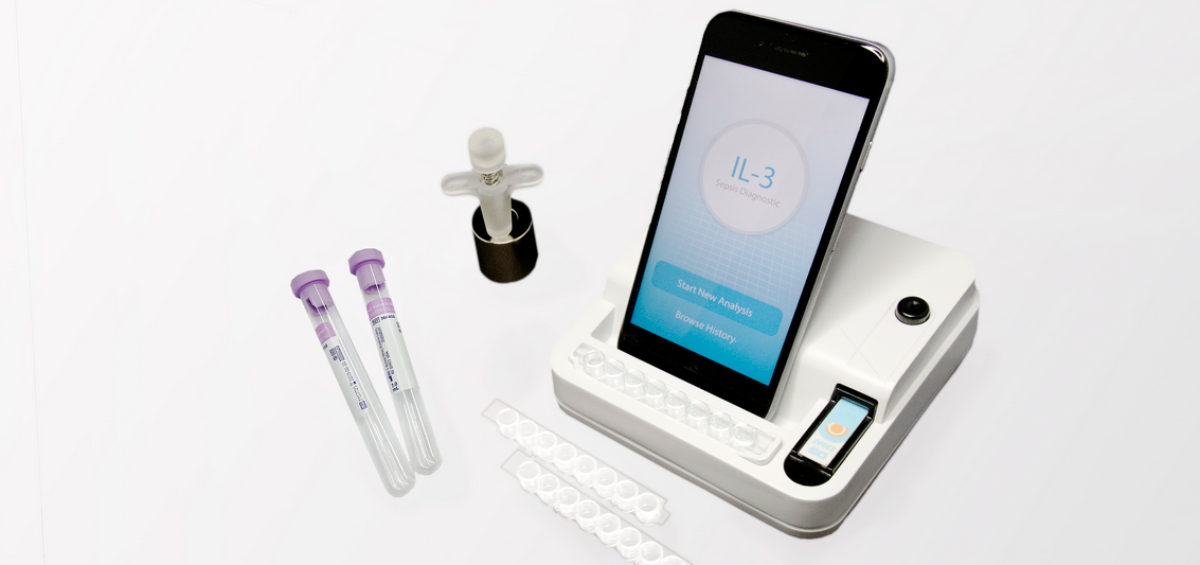
Here, we report the development of a point-of-care (POC) platform for rapid sepsis detection, called IBS (integrated biosensor for sepsis).
2) Can you describe the platform? What does it do and how does it work?
The IBS prototype measures levels of a protein called cytokine interleukin-3 (IL-3) in the blood. IL-3 has recently been found to play a role in sepsis development.
Simply put, the platform uses magnetic beads to directly and quickly extract target protein IL-3 from blood samples. The beads are adorned with antibodies and electron mediators, and through a series of reactions, generate an electrical current which provides an analytical readout of IL-3 levels.
In this clinical study, we applied IBS to detect IL-3 in clinical samples. Blood samples were collected from patients with symptoms of systemic infection, inflammation, or both. We obtained 23 samples from septic patients and another 39 from non-septic patients.
3) What were the results when you tested it?
Applying the IBS prototype, we achieved rapid (<1 hr) and highly sensitive IL-3 detection in human plasma samples. The pilot clinical study (n = 62) supported the potential of IL-3 as an accurate surrogate biomarker of sepsis: 91.3% of patients with sepsis were correctly identified, and 82.4% of patients without sepsis were correctly diagnosed as not having the disease. In comparison, procalcitonin (PCT), another biomarker for sepsis, has been shown to have accuracies below 80%.
Because IBS is fast and requires a small blood sample, it can be readily adopted to track temporal changes of biomarkers.
We can monitor a patient’s blood over time to watch for an increase in IL-3 levels, which would allow us to more quickly identify patients at risk of developing sepsis.
Also, using a small sample would be especially advantageous for detection of sepsis in newborns/infants, as blood samples from preterm infants are limited in volume.
4) How could this platform be used in a clinical setting?
Using clinical samples, we show that high plasma IL-3 levels are associated with high organ failure rate and thus greater risk of mortality, confirming the potential of IL-3 as an early diagnostic biomarker of sepsis.
With further system development (e.g., full automation, data security measures) and rigorous validation studies, this compact and fast IBS platform could be readily integrated into clinical workflows, enabling timely diagnosis and proactive treatment of sepsis.
To learn more about this work, check out the team’s recent paper published in ACS Nano.
About the Mass General Research Institute
Research at Massachusetts General Hospital is interwoven through more than 30 different departments, centers and institutes. Our research includes fundamental, lab-based science; clinical trials to test new drugs, devices and diagnostic tools; and community and population-based research to improve health outcomes across populations and eliminate disparities in care.
Support our Research






Leave a Comment